The power dilemma of the AI era and the need for long-duration energy storage
Driven by the global energy transition and the wave of electrification, the battery industry is expanding at an unprecedented pace. From consumer electronics to electric vehicles, from energy storage power stations to the low-altitude economy, batteries have become the indispensable "energy heart" of modern society. However, another force that cannot be ignored – the explosive evolution of artificial intelligence (AI) – is quietly reshaping the structure of electricity demand, pushing energy storage to a new strategic level.
The higher the intelligence of AI technology, the greater the demand for computing power, and electricity consumption rises sharply as a result. During 2025–2026, AI data centers in the United States will add 6 to 13 gigawatts of electricity demand each year, with peak growth rates reaching four times the historical average. Globally, data center electricity demand is expected to reach 156 GW by 2030, corresponding to an investment scale exceeding $5 trillion. Elon Musk recently proposed a notable energy solution: deploying industrial-scale battery energy storage systems on a large scale to charge the grid at night and discharge during the day to unlock AI demand – a model that could double the overall power generation efficiency of the United States.

Figure 1 power station supporting a high-demand center
The "24/7 uninterrupted operation" characteristic of AI data centers creates a rigid demand for around-the-clock stable power supply. At the same time, there is a significant temporal mismatch between the peak generation periods of renewable energy sources such as solar and wind (midday and windy nights) and the peak electricity demand period (evening). To bridge this "spatiotemporal mismatch," it is necessary to rely on long-duration energy storage (LDES) technologies capable of discharging continuously for more than four hours.
Against this backdrop, flow batteries, with their inherent safety, ultra-long cycle life, and decoupling of power and capacity, are moving from being an "alternative" in energy storage technology to playing a "key role in long-duration storage." In 2025, newly installed flow battery capacity in China increased by 43% year-on-year, and a total of 86 flow battery projects had been put into operation nationwide. The global flow battery market is expected to grow from $830 million in 2025 to $1.41 billion by 2030, at a compound annual growth rate of 12.0%.

Figure 2 renewable energy power station with energy storage system
Flow batteries – the "liquid heart" of energy storage
Flow batteries, similar to lithium-ion batteries, come in many types but have different structures and characteristics.
Members of flow battery family
A flow battery is a new type of rechargeable battery in which energy is stored in electrolyte solutions, and energy storage and release are achieved through the flow of electrolytes and electrode reactions. Unlike traditional lithium-ion batteries, the electrolyte in flow batteries is stored in external tanks, making them easy to scale up, inherently safe, and long-lasting.
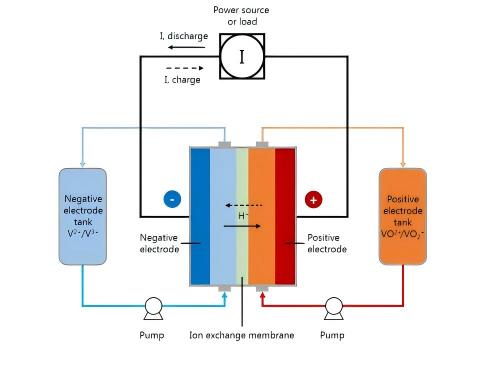
Figure 3 schematic diagram of a vanadium redox flow battery
Based on the different active materials in the electrolyte system, flow batteries can be divided into multiple technology pathways. Among them, the most commercially mature and industrially supported is the vanadium redox flow battery (VRFB). In addition, iron-chromium flow batteries use iron and chromium redox couples as active materials, and zinc-bromine flow batteries use zinc and bromine; both have lower electrolyte costs. Organic flow batteries, with their advantages of low cost, high safety, and molecular designability, have emerged as a new technology route, with the number of related global patents increasing by more than 200% year-on-year in 2025. From a product classification perspective, there are more than 20 technology routes for flow batteries, all of which store and release electrical energy through the mutual conversion of different electrolyte ions.
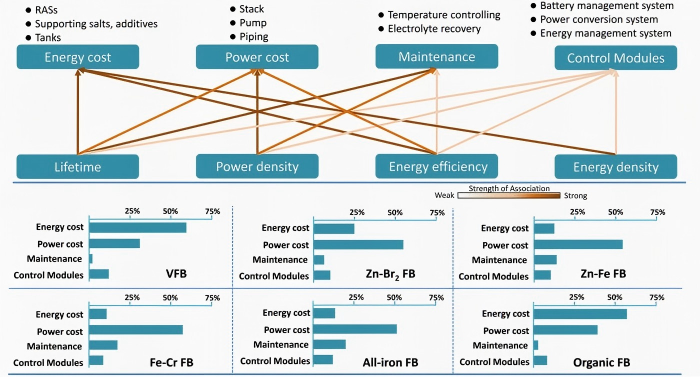
Figure 4 comparison of flow battery technology routes [1]
Principle of flow batteries
The basic structure of a flow battery includes electrolyte storage tanks, pumps, stacks (reactors), membrane electrode assemblies, and electrodes. During operation, the positive and negative electrolytes are pumped from their respective tanks into the stack, where redox reactions occur on the electrodes on either side of the ion-exchange membrane.
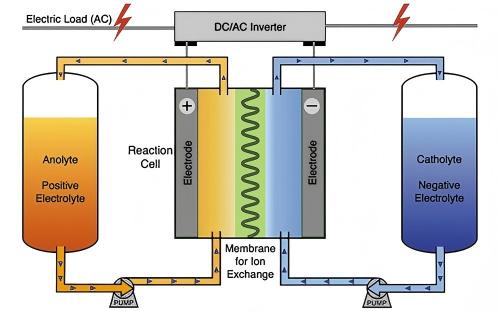
Figure 5 schematic diagram of a flow battery system structure
Taking the most commercially mature vanadium redox flow battery as an example, the electrolyte uses vanadium ions in different valence states as active materials: during charging, V4+ (tetravalent vanadium) in the positive electrode loses electrons to become V5+ (pentavalent vanadium), while V3+ (trivalent vanadium) in the negative electrode gains electrons to become V2+ (divalent vanadium); the reverse reactions occur during discharging. Because the active materials in flow batteries are completely dissolved in the liquid and no phase change occurs during the reaction, they can be deeply charged and discharged, withstand high currents, and have very low self-discharge – almost no self-discharge when the system is in off mode.
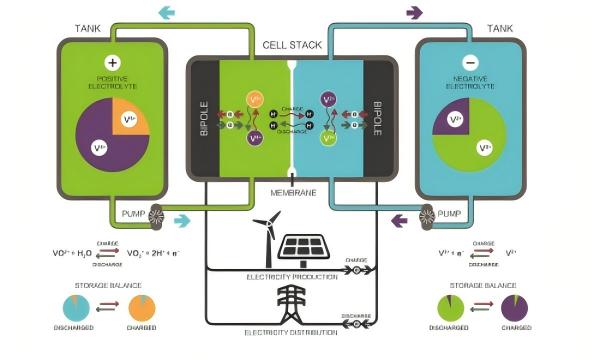
Figure 6 working principle of a vanadium redox flow battery
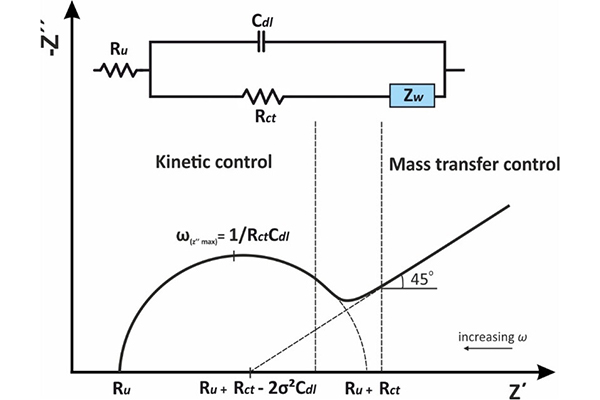
The core design concept of flow batteries is the decoupling of power and capacity. The number and size of stacks determine the output power of the battery, while the volume of the electrolyte determines the energy storage capacity. The two are independent and can be designed separately according to needs. This is fundamentally different from the integration logic of lithium-ion batteries, where "every additional kilowatt-hour of storage requires replicating the entire reaction structure." In addition, flow batteries have the advantage of extremely fast charge-discharge response: they can switch between charging and discharging states in just 0.02 seconds, with a response speed of only 1 millisecond.
Advantages and disadvantages of flow batteries
The advantages of flow batteries in large-scale energy storage scenarios are very prominent.
Safety is their core competitive advantage: flow batteries generally use water-based electrolytes, which are non-flammable, and even if the positive and negative electrolytes mix, they will not catch fire or explode. The vanadium redox flow battery energy storage power station built by Dalian Rongke Energy Storage in 2012 has been operating safely and stably for more than ten years.
Extremely long cycle life: flow batteries can achieve 10,000 cycles, and some technology routes can even reach more than 20,000 cycles, with an overall service life of 20 years or more.
Flexible energy capacity expansion: simply by increasing the capacity of the electrolyte storage tanks, the energy capacity of the battery can be easily expanded.
Good full-life-cycle economics: although the initial investment cost is relatively high, the longer the storage duration of a flow battery, the lower the levelized cost of storage, and the electrolyte can still be recycled after system degradation, resulting in high residual value.
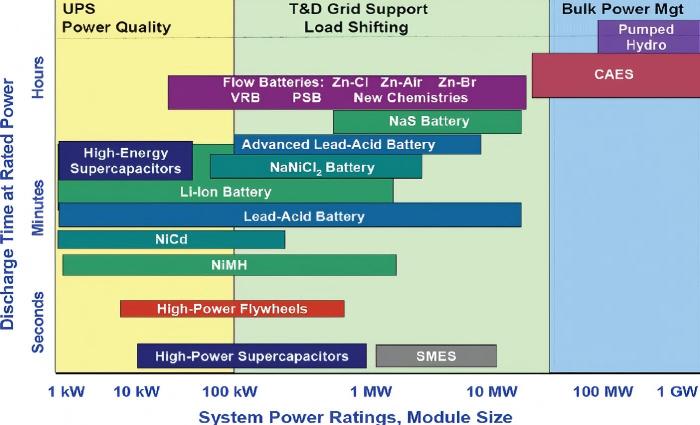
Figure 7 comparison of various battery technologies
Low energy density is the biggest drawback of flow batteries. The energy density of vanadium redox flow batteries is about 25 Wh/L, only about 10% that of lithium-ion batteries, so they require more space. High initial investment cost: the price of vanadium redox flow battery energy storage systems is mainly concentrated in the range of 1.946–2.76 yuan/Wh, with an average price of 2.28 yuan/Wh. High system complexity: they involve additional components such as electrolyte circulation pumps, piping systems, and thermal management, making operation and maintenance relatively complex. In addition, active materials in the electrolyte may cross the separator, leading to capacity degradation – a traditional problem that has been overcome by the latest "balanced state" electrolyte strategy. Batteries using 15-micron-thick Nafion membranes with balanced state electrolyte showed a reduction in capacity fade rate from 0.061% to 0.015% over 1,000 cycles, a decrease of 75.4%.
How to test flow batteries
The current special market for flow batteries is the long-duration energy storage market driven by AI and various overloaded electricity demands. Based on their application characteristics, corresponding battery test items are needed to verify the performance of flow batteries.
Battery performance required for long-duration energy storage
Long-duration energy storage scenarios impose unique performance requirements on batteries that differ from those for electric vehicles. Intrinsic safety is the primary consideration – energy storage power stations are deployed in densely populated areas or near power plants, and once a fire starts, it is extremely difficult to extinguish. Therefore, the water-based electrolyte design of flow batteries gives them a natural advantage in this regard. Ultra-long cycle life determines the full-life-cycle economics of the energy storage system. The cycle life of flow batteries exceeds 10,000 cycles, far superior to the 2,000–3,000 cycles of lithium batteries. Capacity recoverability is an important economic indicator for energy storage systems. Flow battery electrolyte can be replaced online and active materials can be recycled, resulting in low capacity recovery costs. High energy efficiency directly affects the operational economics of the energy storage system. The industry threshold requires energy conversion efficiency to exceed 80%, and currently, the 42 kW vanadium redox flow battery stack from the China Petroleum Engineering Materials Research Institute has achieved an energy conversion efficiency of 83%. Low self-discharge rate is crucial for long-duration storage. Flow batteries have almost no self-discharge when the system is in off mode, and their charge does not decay after being stored for days or even weeks.
How to test the performance of flow batteries
Performance testing of flow batteries needs to cover the entire chain from material level to system level. The core test items include:
Stack performance testing: test rated power, maximum power, current density (industrial applications are concentrated in the range of 160–200 mA/cm2), voltage efficiency, coulombic efficiency, energy efficiency (industry requirement >80%), polarization characteristics, power density, etc.
Electrolyte performance testing: test vanadium ion concentration, valence state distribution, electrolyte utilization rate, active material stability, thermal stability, viscosity and flow characteristics, impurity content, etc.
System-level testing: includes system energy efficiency (total energy conversion efficiency from charging to discharging), capacity retention rate, self-discharge rate, response time (flow batteries can reach millisecond level), cycle life testing (thousands to tens of thousands of cycles), thermal management effectiveness, pump energy consumption, etc.
IEC 62932-2-1 standard testing: the world's first core international standard for flow batteries, led by the Dalian Institute of Chemical Physics, Chinese Academy of Sciences. It specifies general performance requirements and test methods for stationary flow battery energy systems, covering key items such as energy test, maximum input test, maximum output test, and energy efficiency test.
In addition, the "balanced state" electrolyte strategy, as an innovative test and evaluation method, precisely controls transmembrane ion flux by independently adjusting concentration and valence state, effectively verifying the controllability of capacity fade. System-level testing also needs to simulate real application scenarios such as grid peak shaving and renewable energy integration, verifying the BMS's balancing control capability and system reliability.
Necessity of high and low temperature testing
Flow batteries face complex temperature environments in actual operation. In regions with extreme temperature differences such as Xinjiang and Inner Mongolia, energy storage power stations must withstand an annual temperature span from -30℃ to +45℃. High and low temperature testing is crucial for verifying flow battery performance.
Electrolyte rheology: At low temperatures, electrolyte viscosity increases, affecting pumping efficiency; at high temperatures, decomposition of active materials may accelerate. It is necessary to test the change in flow characteristics within the range of -20℃ to 60℃.
Stack performance stability: Temperature changes directly affect the rate constants of redox reactions and the conductivity of the membrane. High and low temperature tests need to verify the consistency of voltage efficiency, coulombic efficiency, and energy efficiency of the stack over a wide temperature range.
Thermal management effectiveness: At high current densities, the internal temperature of the stack rises significantly. It is necessary to verify the heat dissipation capacity and temperature uniformity control of the flow battery's own thermal management system.
Capacity fade mechanism: Extreme temperatures may accelerate the transmembrane migration of active materials and side reactions. High and low temperature cycle testing helps reveal the pattern of capacity fade, providing data support for life prediction models.
High and low temperature testing is not only a necessary step in the development of flow battery products but also a key verification item required by international standards such as IEC 62932-2-1.

Figure 8 NEWARE high and low temperature All-in-One Tester
The Future of flow batteries
Flow batteries are transitioning from demonstration validation to large-scale commercial application. Driven by long-duration electricity demand from AI and data centers at night, the next five to ten years will be a critical period for their large-scale deployment, capacity expansion, and market validation. With breakthroughs in high-power stacks and the maturity of electrolyte leasing models, system costs are continuously decreasing. Applications will focus on renewable energy integration, grid peak shaving, photovoltaic-storage-charging stations, and long-duration storage for commercial and industrial users. The largest vanadium redox flow battery energy storage power station in China – the Xinjiang Jimsar 200MW/1GWh project – was officially put into operation at the end of 2025, making it the world's first single-unit gigawatt-hour-scale flow battery energy storage project. This marks that flow batteries have the engineering capability to support large-capacity, long-duration energy storage needs.

Figure 9 Xinjiang Jimsar 200MW/1GWh flow battery energy storage station
Reference
[1] Zhang C, Yuan Z, Li X. Designing better flow batteries: an overview on fifty years' research[J]. ACS Energy Letters, 2024, 9(7): 3456-3473.
Supplement: Some of the information presented above was obtained from the Internet. We are very sorry if there is any infringement! You can contact us for deletion!