Introduction
On February 26, 2026, a research team from Nankai University and Shanghai Space Power Research Institute published findings in Nature that challenge a 200-year-old assumption in electrolyte design .
Conventional lithium-ion batteries rely on oxygen-based solvents, a paradigm traceable to Alessandro Volta's 1800 invention of the electric pile . The Nankai team's approach? Replace oxygen with fluorine in the solvent molecules.
The results are striking:
| Performance Metric | Conventional Li-ion | Nankai Fluorinated System |
| Room Temperature | 160-300 Wh/kg | >700 Wh/kg |
| -50°C Performance | Typically fails | ~400 Wh/kg maintained |
| Operating Range | -20°C to 45°C | -50°C to room temperature |
Why fluorine? understanding the mechanism
The fundamental limitation of conventional electrolytes lies in the ion-dipole interaction between lithium and oxygen in carbonate solvents . While this interaction enables lithium salt dissolution, it creates several challenges:
Strong coordination hinders interfacial charge transfer kinetics.
Poor wettability requires excessive electrolyte loading.
Low-temperature performance degradation becomes severe below -30°C.
The Nankai team's fluorinated hydrocarbon solvents replace oxygen with fluorine, fundamentally altering this interaction:
"We design a series of novel fluorinated hydrocarbon solvent molecules to replace oxygen with fluorine, successfully constructing a weak coordination electrolyte for lithium metal batteries" .
The benefits stem from weaker Li-F coordination, which:
- Accelerates charge transfer kinetics, particularly at low temperatures.
- Improves electrode wettability, reducing electrolyte consumption.
Enables tunable electron density through molecular design.
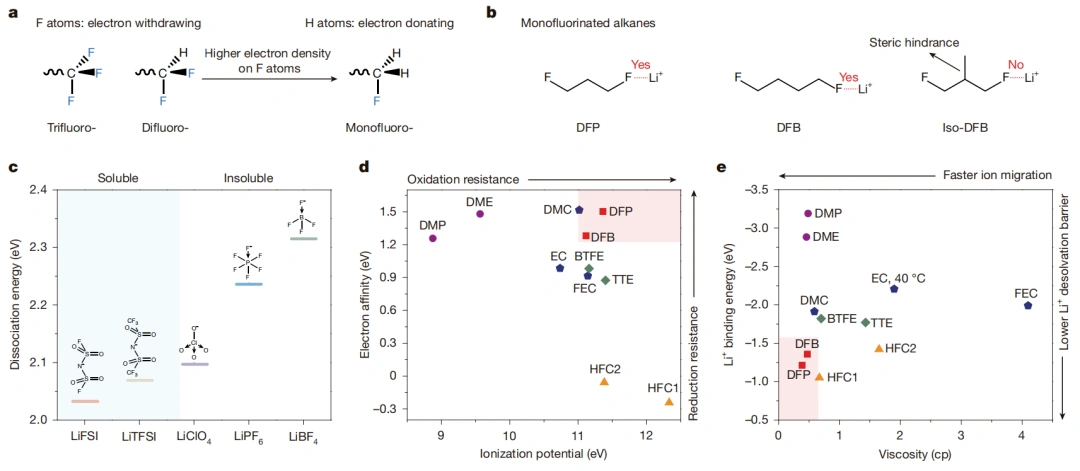
Figure 1. Design principles and characteristics of hydrofluorocarbons as electrolyte solvents.
The reproducibility challenge
Professor Chen Jun, corresponding author and academician of the Chinese Academy of Sciences, emphasizes that translating laboratory breakthroughs into practical technologies requires rigorous validation :
"We can't always stay in the ivory tower. Our goal is to address real industrial challenges."
This transition from discovery to application raises critical questions for researchers attempting to replicate or build upon these findings:
Key validation questions
- Signal Integrity: How do researchers distinguish genuine electrochemical behavior from measurement artifacts when analyzing novel electrolyte systems?
- Temperature Control: When testing at -50°C, how does one ensure that performance data reflects material properties rather than environmental fluctuations?
Reproducibility: What equipment specifications enable consistent results across different laboratories?
Measurement requirements for advanced electrolyte research
Validating fluorinated electrolyte systems requires instrumentation capable of capturing subtle electrochemical signatures across extreme temperature ranges.
Critical specifications
| Parameter | Requirement | Rationale |
| Current/Voltage Accuracy | ±0.01% FS or better | Captures minor side reactions during SEI formation |
| Response Time | ≤10 ms | Resolves fast charge transfer kinetics |
| Temperature Uniformity | ±0.5°C across chamber | Ensures consistent低温performance data |
| Channel Density | 8-128 channels | Enables statistical validation |
Instrumentation considerations
When selecting equipment for advanced electrolyte research, several factors influence data quality and publication readiness.
Accuracy and precision
The Nankai study involves characterizing electrochemical behavior across a 100°C temperature range. Maintaining measurement integrity under such conditions requires:
Low-noise electronics that don't introduce artifacts at extreme temperatures.
Thermal compensation circuits that maintain calibration across temperature swings.
High-resolution ADCs (≥24-bit) for capturing subtle voltage plateaus.
Environmental control
Testing at -50°C presents unique challenges beyond simply cooling the cell:
Frost prevention without affecting gas exchange.
Mechanical isolation to prevent vibration-induced contact loss.
Ramp rate control to avoid thermal shock to materials.
Data management
Modern battery research generates terabytes of data. Effective validation requires:
Synchronized logging of electrical and environmental parameters.
Automated analysis routines for identifying outliers.
Export compatibility with common plotting and analysis tools.
Practical implications for battery laboratories
For research groups working on next-generation electrolytes—whether fluorinated hydrocarbons, solid-state systems, or sodium-ion chemistries—the equipment behind published results matters.
For academic researchers
When preparing manuscripts for journals like Nature, Joule, or the Journal of The Electrochemical Society, reviewers increasingly scrutinize measurement methodology. Key questions include:
How were temperature gradients characterized during low-temperature testing?
What calibration procedures ensured accuracy across the measurement range?
How many parallel channels confirmed statistical significance?
For industrial R&D teams
For companies developing commercial cells based on emerging chemistries, production-scale validation requires:
Scalable testing architectures that transition from coin cells to pouches.
Traceable calibration for quality system compliance.
Data integrity features supporting regulatory submissions.
Case study: validating low-temperature performance
Consider a researcher attempting to verify the -50°C performance claims from the Nankai study. The experimental protocol involves:
Cell preparation using fluorinated electrolyte formulations.
Temperature stabilization at -50°C for ≥4 hours.
Discharge testing at various C-rates.
Data analysis comparing capacity retention to room-temperature baseline.
Potential pitfalls
Without appropriate instrumentation, several artifacts can compromise results:
| Pitfall | Consequence | Prevention |
| Thermal gradients | Non-uniform electrode utilization | Multi-point temperature monitoring |
| Contact resistance | Misleading impedance data | Four-wire sensing |
| Channel crosstalk | Apparent capacity variations | Electrically isolated channels |
| ADC quantization | Missed voltage plateaus | ≥24-bit resolution |
Industry context: the path to commercialization
Professor Chen's team has already demonstrated practical applications of their technology, collaborating with automaker Hongqi to release a mass-producible ultrahigh energy density lithium-rich manganese solid-liquid battery system exceeding 500 Wh/kg . Vehicles equipped with these batteries are expected to enter mass production by late 2026 .
This rapid translation from Nature paper to production vehicle underscores an important trend: the gap between laboratory discovery and commercial application is narrowing.
For equipment providers, this means supporting researchers through multiple validation phases:
Phase 1: Coin cell screening for electrolyte formulations.
Phase 2: Pouch cell validation under realistic conditions.
Phase 3: Production-scale formation and grading.
Selection criteria for battery test equipment
When evaluating instrumentation for advanced electrolyte research, consider these factors:
Technical specifications
Accuracy: Look for ±0.02% FS or better for research applications.
Rise Time: ≤10 ms for capturing fast kinetics.
Temperature Range: -70°C to 200°C for comprehensive characterization.
Channel Count: Scalable from 8 to 512 channels.
Software capabilities
Programmability: Flexible step definitions for complex protocols.
Real-time visualization: Immediate feedback during long-term cycling.
Export formats: CSV, Excel, and direct plotting tool integration.
Support and validation
Calibration traceability: NIST or equivalent standards.
Application support: Technical assistance for unusual protocols.
Installation qualification: Documentation for QA/QC compliance.
Conclusion
The Nankai University fluorinated electrolyte breakthrough represents a significant advance in battery technology, potentially enabling electric vehicles that perform reliably in extreme climates and aerospace applications requiring high energy density at low temperatures .
However, translating such discoveries from published papers to reproducible laboratory results—and eventually to commercial products—depends critically on measurement quality.
As battery research pushes toward 700 Wh/kg and beyond, the relationship between chemical innovation and measurement precision becomes increasingly symbiotic. Breakthroughs in materials demand commensurate advances in characterization capabilities.
About NEWARE
Our CT-4008 series, featuring ±0.01% FS accuracy and 24-bit resolution, coupled with integrated thermal chambers operating from -70°C to 150°C, provides unwavering data integrity for every experiment pushing toward the 700 Wh/kg limit.
For researchers working on next-generation electrolytes, NEWARE offers:
CT-4008 Series: Featuring ±0.01% FS accuracy, ≤1 ms response time, and a 10 Hz data logging frequency to precisely capture rapid charge transfer kinetics.
Integrated Thermal Chambers: -70°C to 150°C with uniform temperature fields.
High-Channel-Count Systems: 128+ parallel channels for statistical validation.
BTS 9.0 Software: Advanced analysis tools for impedance, dQ/dV, and cycle life.
References
Nankai University research team, Nature, February 26, 2026
Chen, J. et al., "Fluorinated Hydrocarbon Electrolytes for High-Energy Lithium Metal Batteries," Nature, 2026
Industry standard comparison data, battery industry reports, 2025-2026
Chen Jun, academician interview, Chinese Academy of Sciences, February 2026
Hongqi automotive press release, "Mass-Producible Ultrahigh Energy Density Battery System," January 2026
Frequently Asked Questions
Q: What accuracy is needed for validating novel electrolyte systems?
A: For research-grade validation of fluorinated or other advanced electrolytes, ±0.01% to ±0.02% FS accuracy is recommended to capture subtle voltage plateaus and side reactions.
Q: How to ensure the reproducibility of low-temperature test results?
A: Key factors include temperature uniformity (±0.5°C or better), adequate soak times (≥4 hours at target temperature), and synchronized logging of thermal and electrical parameters.
Q: Can the same equipment handle coin cells and pouch cells?
A: Look for systems with multiple current ranges (mA to A) and flexible fixturing that accommodates various form factors without sacrificing accuracy.
Q: What software features accelerate electrolyte research?
A: Automated data fitting for impedance analysis, dQ/dV curve generation, and batch processing capabilities significantly reduce analysis time for high-channel-count experiments.











