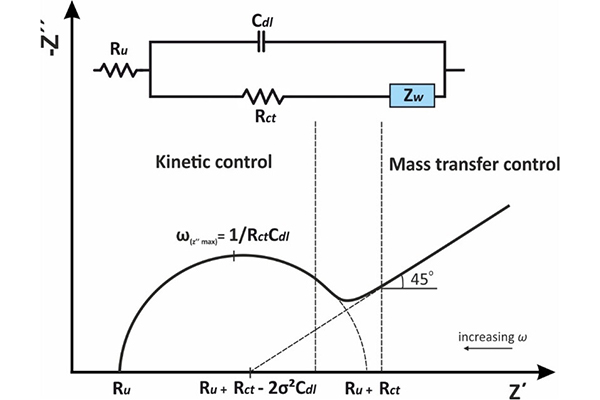
Introduction
Coupling Cyclic Voltammetry (CV) with in situ testing techniques is a core strategy for gaining a deep understanding of battery behavior. Unlike ex situ testing, in situ testing coupled with CV can be performed synchronously, acquiring other signals in real-time during CV measurement. This establishes real-time, direct causal relationships and reveals the dynamic evolution processes inside batteries.
Advanced in situ coupled testing
In situ testing represents the forefront of research directions. It enables the synchronous acquisition of other physical or chemical signals from the same battery at the same time as the CV test, thereby establishing the most direct causal relationships.
In situ X-Ray diffraction
Coupling logic: CV (Electrochemical signals) + XRD (Crystal structure signals)
Synergistic effect: CV is responsible for applying precisely controlled electrochemical perturbations and monitoring the current-voltage response in real-time. In situ XRD probes the same battery simultaneously during the CV test, analyzing the evolution of the material's internal crystal structure at that moment. The advantage of in situ XRD lies in its ability to observe the evolution of the electrode material's crystal structure in real-time (such as lattice parameter changes, generation and disappearance of new phases). It can directly correlate each redox peak on the CV curve with specific structural phase transitions, which is crucial for studying the phase transition mechanism of electrode materials.
In situ spectroscopy
Coupling logic: CV (Electrochemical signals) + Spectroscopy (Molecular/chemical bond signals)
Synergistic effect: The coupling of CV with in situ spectroscopy links the macroscopic electrochemical response to microscopic molecular structural changes. It can not only directly identify reaction intermediates or active substances generated at specific potentials but also analyze complex reaction pathways, providing powerful assistance for evaluating material and interface stability.
Researchers from the Shanghai Synchrotron Radiation Facility utilized Quick-scanning X-ray Absorption Fine Structure (QXAFS) technology coupled with CV to track the reconstruction process of two cobalt-based catalysts under oxygen evolution reaction conditions in real-time with millisecond-to-second-level temporal resolution (Figure 1) [1].
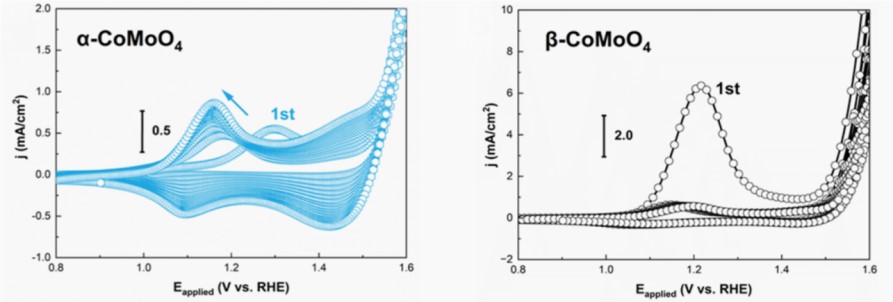
Figure 1 consecutive CV scans tracked the electrochemical responses of α-CoMoO4 and β-CoMoO4 in 1 M KOH
The study found that while both catalysts ultimately reconstructed into the active CoOOH phase, their transformation pathways and rates were distinctly different. Notably, β-CoMoO4 demonstrated exceptional performance by completing a rapid and comprehensive reconstruction within a single CV cycle (approximately 6 minutes) (Figure 2).
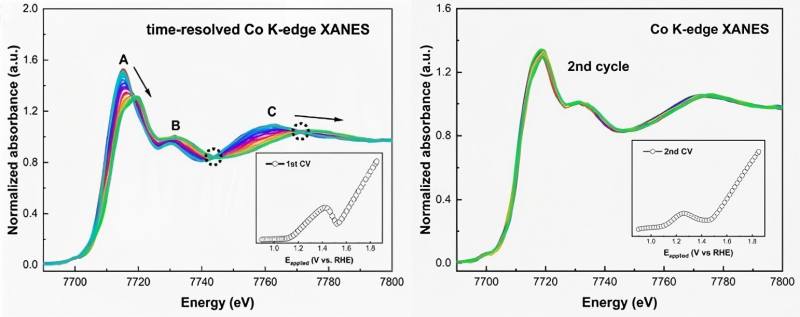
Figure 2 time-resolved operando Co K-edge XANES spectra of β-CoMoO4 during the (left) first and (right) second cyclic voltammetry cycles between 0.9V and 1.85V. Insets show the corresponding cyclic voltammetry curves
Considering the influence of internal resistance from both the electrode and electrolyte, researchers also collected linear sweep voltammetry (LSV) curves with iR compensation (Figure 3).
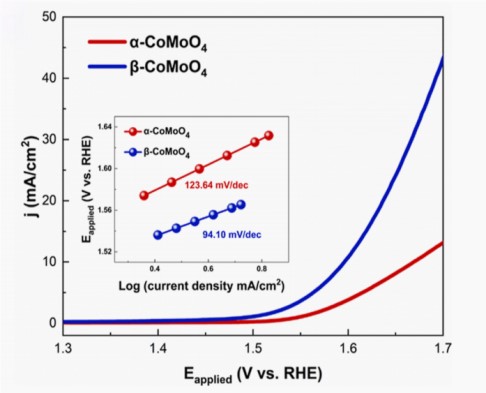
Figure 3 the polarization curves of α-CoMoO4 and β-CoMoO4 in 1 M KOH at a scan rate of 2 mV/s, after iR correction
The inset displays the corresponding Tafel plots
By combining QXAFS data with CV data, the researchers not only elucidated the changes in atomic structure but also identified that the large number of μ2-OH-Co2+/3+ active sites generated after reconstruction are key to the performance enhancement. This work clearly demonstrates how to precisely correlate the electrochemical responses in CV with real-time atomic-level structural changes of the catalyst.
In addition, the combination of in situ Raman spectroscopy and CV testing can identify molecular structural changes of substances on the electrode surface in real time and track the formation of reaction intermediates. The combination of in situ Fourier transform infrared spectroscopy and CV testing is particularly suitable for studying the chemical composition at the electrode/electrolyte interface, such as electrolyte decomposition and the formation of the SEI film. These spectroscopic techniques can directly answer the question, "What new substances are generated at this CV potential?"
In situ microscopy techniques
Coupling logic: CV (electrochemical signals) + Microscopy (morphological signals)
Synergistic effect: In situ scanning electron microscopy can observe the microstructural changes of electrode materials during charge and discharge processes in real time, such as volume expansion, crack formation, and lithium dendrite growth. Research from institutions such as Beijing University of Technology, using in situ electrochemical scanning electron microscopy, directly revealed the dynamic process of lithium dendrite growth in solid-state batteries and its hazards (Video1, Video 2) [2]. The study not only visualized dendrite growth but also found that higher current densities (0.05 mA cm-2) induce more significant mechanical stress, leading to electrolyte tearing, while lower current densities (0.01 mA cm-2) facilitate dendrite dissolution and crack closure. Based on these observations, the researchers established an electrochemical-mechanical stress coupling model, providing critical theoretical foundations for designing and producing safer solid-state batteries (Figure 4).
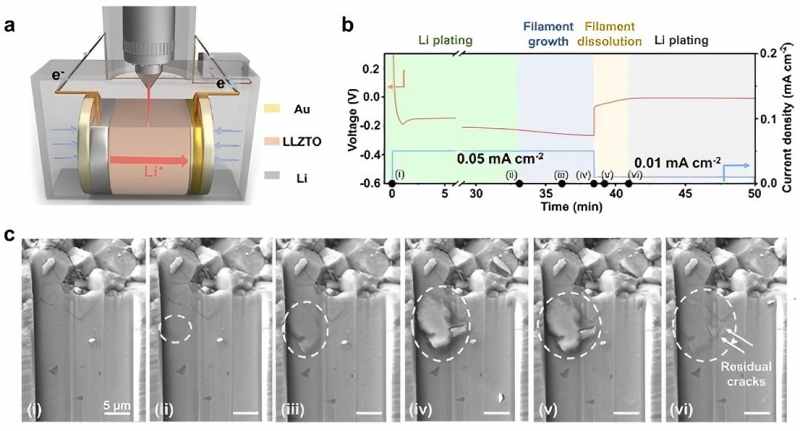
Figure 4 (a) experimental setup for in situ observation of lithium dendrite growth within the SSE, (b-c) current-voltage changes in the electrochemical curve and the corresponding cross-sectional morphological evolution of the inorganic solid-state electrolyte LLZTO observed via in situ SEM

Figure 5 schematic diagram of the coupled system integrating in situ electrochemical scanning electron microscopy with the NEWARE high-performance battery testing system
Video 1 In situ observations of the growth and dissolution of Li filamentsn
Video 2 CCD reduction caused by the irreversible mechanical damage of the SSE
In situ liquid cell transmission electron microscopy: Holtz et al. developed a quantitative electrochemical method that utilizes liquid cell transmission electron microscopy (TEM) coupled with electron energy loss spectroscopy (EELS) to image ion distributions at the microstructural level and observe local electronic structure changes at the nanoscale during operation [3]. The specialized application of a flow cell to valence energy-filtered transmission electron microscopy (EFTEM) facilitated the probing of low-energy states (1-10 eV), enabling the tracking of ion dynamics during battery charging and discharging, specifically the transfer of ions from the electrode to the electrolyte. Based on ab initio nonlinear response theory, they tracked the electronic structures of both solvated ions and intercalated ions. During the real-time charging/discharging process, the state of Li in a LiFePO4 electrode and an aqueous electrolyte was determined with nanoscale resolution (Figure 6). Simultaneously, Li transfer between the electrolyte and the electrode, as well as the charging kinetics, were tracked. Under the same conditions, competitive delithiation mechanisms were observed.
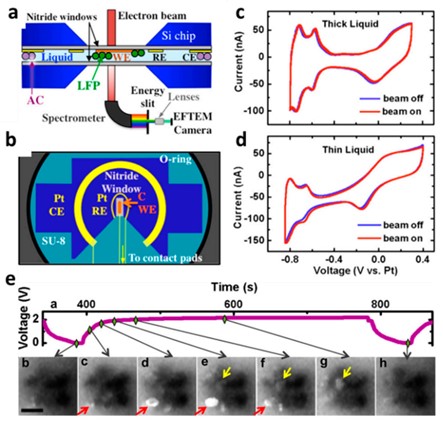
Figure 6 schematic diagram of the TEM holder cross-section: (a) Tubular silicon nitride membrane encapsulating the fluid layer and the in situ electrochemical workstation, (b) Patterned three-electrode system on the top chip, (c, d) Electrochemical activity of Pt in cyclic voltammetry (CV) with a liquid layer thickness of approximately 150 nm, (e) Time evolution (a-h) of LiFePO4/FePO4 clusters during charge/discharge cycles. The red and yellow arrows indicate the core-shell structure and the propagation of the delithiation process along the path from left to right, respectively.
Conclusion
The in situ testing techniques discussed above enable direct correlation of electrochemical behavior with macroscopic physical structural changes. By selecting appropriate in situ methods to complement CV measurements, researchers can gain profound insights into structural transformations induced by internal battery reactions from multiple perspectives. This approach provides valuable guidance for subsequent studies.
References
[1] Zhong X, Hou C, Chen Y, et al. Unraveling the Contrasting Dynamics of Reconstruction in Wolframite Cobalt Molybdate Polymorphs for Oxygen Evolution Reaction Electrocatalysis[J]. ACS Catalysis, 2025, 15: 11958-11969.
[2] Cao T, Xu R, Cheng X, et al. Chemomechanical origins of the dynamic evolution of isolated Li filaments in inorganic solid-state electrolytes[J]. Nano Letters, 2024, 24(6): 1843-1850.
[3] Holtz M E, Yu Y, Gunceler D, et al. Nanoscale imaging of lithium ion distribution during in situ operation of battery electrode and electrolyte[J]. Nano letters, 2014, 14(3): 1453-1459.